 In the mid-19th century, a German physicist named Rudolph Clausius, one of the founders of the concept of thermodynamics, was working on a problem concerning efficiency in steam engines and invented the concept of entropy to help measure useless energy that cannot be converted into useful work. And if there are more chimps.īut don't feel bad if you're confused: the definition can vary depending on which discipline is wielding it at the moment: Entropy concerns itself more with how many different states are possible than how disordered it is at the moment a system, therefore, has more entropy if there are more molecules and atoms in it, and if it's larger. So, if you were to look at two kitchens - one very large and stocked to the gills but meticulously clean, and another that's smaller with less stuff in it, but pretty trashed out by chimps already - it's tempting to say the messier room has more entropy, but that's not necessarily the case. Of course, the entropy depends on a lot of factors: how many chimpanzees there are, how much stuff is being stored in the kitchen and how big the kitchen is. 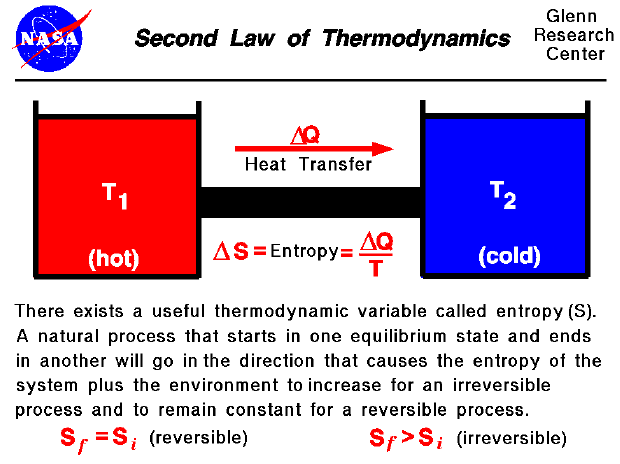
It has more to do with how many possible permutations of mess can be made in that kitchen rather than how big a mess is possible. However, entropy doesn't have to do with the type of disorder you think of when you lock a bunch of chimpanzees in a kitchen. It's harder than you'd think to find a system that doesn't let energy out or in - our universe is as good an example of one as we have - but entropy describes how disorder happens in a system as large as the universe or as small as a thermos full of coffee. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
